The D Vitamins and Their Precursors
It must be recognized that there is not a single vitamin D compound but that there is a family of compounds that exhibit vitamin D activity. The most important of the vitamin D compounds are vitamins D2 and D3 (Figure 1), and the more important of these is vitamin D3 or cholecalciferol. The basis for this statement is that vitamin D3 is the vitamin produced in the skin by ultraviolet irradiation. Vitamin D2, however, is produced by ultraviolet irradiation of the plant sterol ergosterol. The photolysis reaction that converts the 5,7-diene sterols to the corresponding D vitamins has been examined intensively. It is known that 250-310 nm ultraviolet light brings about the photolytic conversion of vitamin D to the previtamin D. Further activation by light photons brings about the formation of tachysterol, a relatively unstable irradiation product, and lumisterol. The previtamin D is in thermal equilibrium with vitamin D3 and upon heating will rapidly convert to the vitamin D structure. The equilibrium is temperature dependent, and the relative concentrations of the forms of vitamin D at equilibrium are a function of temperature. At room temperature the conversion of the previtamin to the vitamin is slow, and even at skin temperatures it is a very slow process.

Figure 1: Structures of the D vitamins

Figure 2: The sterol precursors of the most common of the known vitamin D substances
There are other forms of vitamin D known (Figure. 1) and their precursors are shown in Figure. 2. At present, the most significant of these minor forms is vitamin D4 , which was synthesized as an analog of vitamin D2 by Windaus and Trautman. This form of vitamin D is a chemical and biochemical curiosity and has been synthesized once again in recent years. In mammals its biological activity is only two-thirds or three-fourths that of vitamin D2 or vitamin D3, whereas vitamin D2 and vitamin D3 are approximately equipotent. In birds, however, vitamin D2 is one-tenth as active as vitamin D3 in every respect. Curiously, New World monkeys show similar discrimination against the vitamin D2 compounds. It is indeed possible that in the future, subtle differences may well be found between vitamin D2 and vitamin D3 in other mammals as well. A basis for discrimination against the vitamin D2 compounds in birds will be discussed later. A detailed discussion of the chemical characteristics of the D vitamins including their mass spectra, nuclear magnetic resonance spectra, etc. has been published elsewhere and interested readers are directed to those sources.
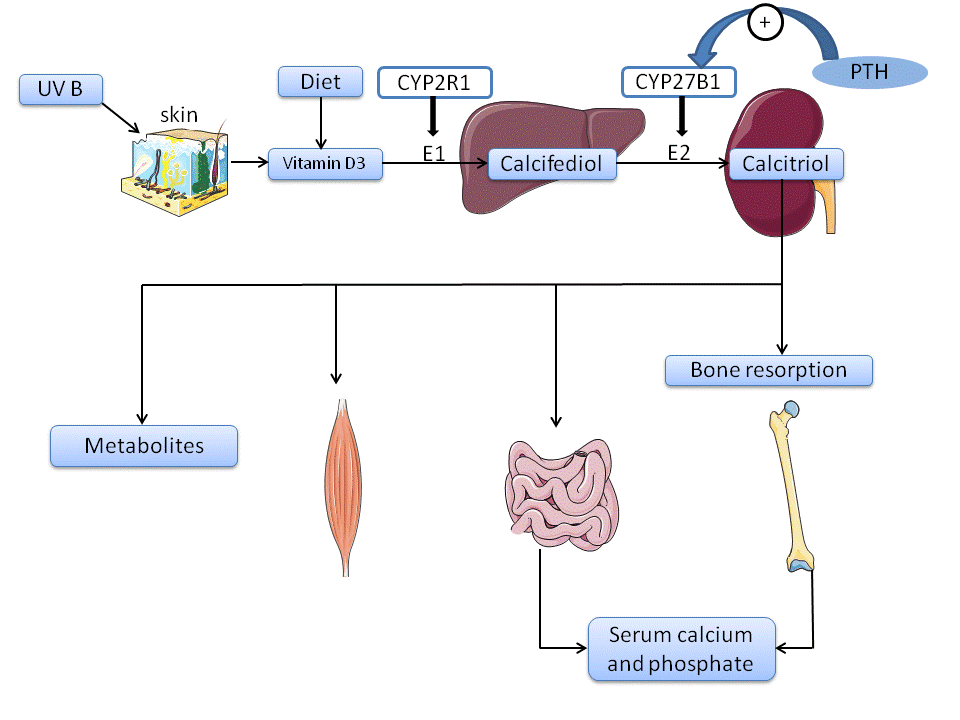
Sources of vitamin D
Several sources of vitamin D include sun exposure, natural diet foods, fortified foods, and supplements. Te best way to obtain vitamin D is sun exposure. Sun exposure enables UVB rays to reach exposed skin to produce vitamin D3. Factors that affect synthesis include the season, time of day, duration of exposure, pollution, skin pigmentation, and sunscreen use. Researchers recommend sun exposure of at least 15 to 30 minutes, twice a week. Example actions include increasing the amount of time exposed to sunlight or the type of clothing worn.
Clinical application of vitamin D compound
1. Rickets, Osteomalacia, and Vitamin D insufficiency
Although many of the hallmarks of rickets and osteomalacia are successfully relieved by doses of vitamin D in the range of 600-800 IU/day (15-20 µg/day) recommended in the 2011 Insitute of Medicine (IOM) report, critics point to epidemiological data that suggest that the current recommended dietary reference ranges (also known as DRIs) do not result in plasma 25-OH-D levels >40 ng/mL, which correlate with maximal bone mineral density or the other health benefts of vitamin D. Consequently, there has been much recent debate over the optimal level of vitamin D intakes, and this has led to a view among some experts that vitamin D intakes might need to be increased above 1500 IU/day and possibly higher in order to achieve target plasma 25-OH-D levels >40 ng/mL. However, the IOM questions the scientifc basis for these claims and points to the fact that recommendations for general populations should be based on current evidence and not opinions.
2. Osteoporosis
As a consequence, it not surprising that vitamin D and vitamin D analogs have been tried in attempts to slow down bone loss and reduce fracture rates in elderly patients with osteopenia and osteoporosis. Small doses of vitamin D (800-1000 IU) have proven effective in treating vitamin D defciency and osteopenia in elderly populations, especially in combination with supplemental calcium, by increasing bone mineral density and reducing fracture rates. However, the use and effectiveness of active vitamin D metabolites in the treatment of osteoporosis remains controversial. Clinical trials of 1α-OH-D3, 1α-OH-D2, and 1α,25-(OH)2D3 have been undertaken in this condition. The experience seems to have been that while benefts have been observed in terms of reductions in vertebral deformities in the second and third years of longer studies, there is no evidence that vitamin D analogs offer advantages over vitamin D or calcium in the treatment of osteoporosis.
3.Secondary hyperparathyroidism and renal osteodystrophy
Chronic renal disease-mineral and bone disorder (CKD-MBD) is accompanied by the gradual loss of renal 25-OH-D3-1α-hydroxylase (CYP27B1) activity over the five-stage natural history of the disease, which culminates in dialysis (stage 5D). Discussion of the optimal vitamin D therapy of CKD patients has refocused attention on the underlying mechansisms for the decline of the renal 1α-hydroxylase activity in this condition. For three decades, it has been widely assumed that the serum 1α,25-(OH)2D3 declines because of enzyme loss in the proximal tubular cells. Recent progress, including the elucidation of the role of fibroblastlike growth factor 23 (FGF-23) in the phosphate homeostatic loop, has shed new light on the role of increasing PO4 and FGF-23 levels in causing dysfunctional vitamin D metabolism in the course of renal disease. Since the known biological effects of FGF-23 include downregulation of the renal 1α-hydroxylase (CYP27B1) and up-regulation of the catabolic 24-hydroxylase (CYP24A1), the possibility exists that the rising FGF-23 level contributes to the fall in serum 1α,25-(OH)2D3 by reducing its synthesis and increasing its degradation. Such a scenario opens the door to the use of CYP24A1 inhibitors in renal disease.
Hyperproliferative conditions: psoriasis and cancer
The demonstration that 1α,25-(OH)2D3 is an antiproliferative prodifferentiating agent for certain cell types in vivo, and many cell lines in vitro suggested that vitamin D analogs might offer some relief in the treatment of hyperproliferative disorders such as psoriasis and cancer. Early psoriasis trials with systemic 1α,25-(OH)2D3 were moderately successful but plagued with hypercalcemic side effects. Modifcations to the protocol included the following:
1.Administration of calcitriol overnight when intestinal concentrations of [Ca2+] were low
2.Substitution of “low-calcemic”analogs for the calcitriol
Immunosuppression
The immunosuppressive properties of 1α,25-(OH)2D3 and its analogs have been the subject of several excellent reviews. 1α,25-(OH)2D3 is believed to work by regulation of the expression of various cytokines, particularly suppressing proinflammatory cytokines and promoting other cytokines, thereby raising the Th2/Th1 ratio. The hormone also stimulates the innate immune system by promoting the transcription of a natural bacterial peptide, cathelicidin (LL-37), which kills Mycobacterium tuberculosis, resulting in increased resistance to tuberculosis. The spectrum of effects exhibited by 1α,25-(OH)2D3 and its analogs on the immune system results in benefcial effects on a wide variety of autoimmune diseases.
The connection of vitamin D with cardiovascular disease does not end with its role in hypertension. Work with various uremic rodent models in which there is secondary hyperparathyroidism and accelerated vascular calcifcation suggests that there are protective effects of calcitriol (and/or 25-OH-D) on the health of the vascular system by suppression of the inflammatory process involved in the development of atherosclerosis, by antiproliferative effects on myocardial cell hypertrophy, and by direct suppressive effects on vascular epithelial cell gene expression of calcifcation genes, e.g., osteocalcin, Runx-2, and osterix. Thus, the emerging concept is that, rather than accelerating renal failure, vitamin D analogs when used at clinically relevant doses are protective to the vasculature and not deleterious, as once believed.
References:
Gombart, A. F. (2012). Vitamin D: Oxidative Stress, Immunity, and Aging. CRC Press.
